Capsules: A Comprehensive Study About Enclosed Drugs
Capsules are solid unit dosage form of medicament in which the drug(s) is enclosed in a practically tasteless, hard or soft soluble container or shell made up of a suitable form of gelatin.
In other word, Capsules are solid dosage forms in which one or more drugs and inert substances are enclosed within a small shell or container generally prepared from a suitable form of gelatin.
Capsules are available in two types:
1. Hard Gelatin Capsules
2. Soft Gelatin Capsules

Hard gelatin capsules are used for filling the solid substances whereas soft gelatin capsules are used for filling the liquids and semisolids.
Advantage
1. Easy to handle and carry
2. Capsules are much economical
3. They are attractive in appearance
4. Better bioavailability and faster onset of action than tablets
5. More stable and have longer shelf life than liquid dosage form
6. Capsules are tasteless, odorless and can easily be administered
7. Have “prescribing flexibility” as a variety of dosage strengths are available
8. The capsule shells can be opacified with titanium dioxide or colored substances, to give protection from light.
9. Packaging and shipping can be done by the manufacturers at a lower cost and less breakage than liquid dosage forms
Disadvantage
1. Hygroscopic drugs not suitable for filling into capsules as they absorb water present in capsule shell. As a result, the shell became very brittle & ultimately lead to crumble into pieces.
2. The concentrated solutions which require previous dilution are unsuitable for capsules because if administered as such lead to stomach irritation
3. Capsules are not suitable for effervescent or deliquescent materials. Efflorescence softens the capsule and deliquescent can dry the capsule shell and make it brittle.
Gelatin
Gelatin is heterogeneous product derived by hydrolytic extraction of animal’s collagen.
Types of Gelatin
There are two basic types of gelatin;
1. Type A
2. Type B
These types are described below;
Type A
Gelatin is mainly derived from pork skin by acid treatment. This process exhibits an iso electric point at pH 9.
Type B
Gelatin is derived from bones and animal skin by alkaline treatment. This process exhibits an iso electric point at pH 4.7.
Why gelatin is the major component of the capsule!
Gelatin is the major component of the capsule. The reason for this is that gelatin possesses five basic properties. They are;
1. Non-toxic
2. It is a good film-forming material
3. Soluble in biological fluids at body temperature
4. Solutions of high concentration, 40% w/v, are mobile at 50°C
5 A solution in water changes from a sol to a gel at temperatures only a few degrees above ambient.
General Characteristics of Gelatin
1. Bloom value
It is a measurement of the gelling power and the strength of the resulting gel. Gelatin generally falls between 50 and 300 bloom strength.
2. Gelling power
It is the key function of gelatin. The gelling power varies depending on the grade of gelatin. It’s measured in terms of bloom value.
The higher the bloom value, the greater the ability to form gel.
3. Viscosity
In general the higher the bloom, the greater the viscosity. Beef bone grades tend to have higher viscosity.
Viscosity is determined by measuring the flow time of a 6.67% solution of gelatin through a U-tube viscometer at 60 °C.
Viscosity measurement is given in millipoise units and varies between 20 and 70mps.
4. Foamability
In general pigskin grades tend to have better foaming properties. Gelatin lowers the surface tension, allowing for the incorporation of air, and stabilizes the foam once aerated.
5. Melting point
Lower melting point gelatins dissolve faster in the mouth, therefore releasing the flavors more quickly for an instant taste sensation.
Lower bloom grades tend to have lower melting points.
6. Color and odor
The gelatin should be as clear as possible and without odor in the solution. Clarity is measured using a turbidimeter.
7. Conductivity
It is a key parameter in photographic applications and grades of the highest purity with minimal or no conductivity are required.
De-ionized grades have an advantage because of their low conductivity.
8. pH
pH is measured using a pH meter on a 1% solution. The pH of gelatin used for capsules ranges from 4 to 6.8.
Preparation of Gelatin

1. Hard Gelatin Capsules
Hard gelatin capsules consist of two cylindrical pieces closed at one end. The shorter part is called the cap. This cap fits over the open end of a longer section called the body. The drug is placed in the body and the cap is slid through, thus encapsulating the drug.

Sizes of Hard Gelatin Capsules
Size | Capacity in mg | Volume in ml |
| 000 | 950 | 1.37 |
| 00 | 650 | 0.95 |
| 0 | 450 | 0.68 |
| 5 | 300 | 0.50 |
| 4 | 250 | 0.37 |
| 3 | 200 | 0.30 |
| 2 | 150 | 0.21 |
| 1 | 100 | 0.15 |
Raw Materials Used for Hard Gelatin Capsules
1. Gelatin
2. Sugar
3. Water 12 to 16 %, may vary depending on the storage condition
4. FD & C and D & C colorant
5. Sulfur dioxide (15%), prevent decomposition during manufacture
6. Opacifying agent: There are two types a) water soluble dyes. For example; erythrosine and b) pigments. For example; iron oxides, titanium dioxide
7. Excipients such as, Preservatives, Diluents, Lubricants and Glidants, Disintegrating agents etc.
Some excipients and their examples are given below;
a) Diluents and fillers: These may be added to the formulation to produce the proper capsule fill volume. For example: Lactose, Microcrystalline cellulose, Starch
b) Disintegrates: These added to the formulation to assist the breakup and distribution of the capsule’s contents in the stomach. For example: Pregelatinized starch, Croscarmellose, and Sodium starch glycolate.
c) Lubricant or glidant: These added to the formulation to enhance the flow properties of the powder. For example: Fumed silicon dioxide, Magnesium stearate, Calcium stearate, Stearic acid, or talc
d) A surface-active agent: They are used to facilitate wetting by the GI fluids. For example: sodium lauryl sulfate.
e) Desiccant materials: They are used to protect capsules against absorption of atmospheric moisture. For example: Dried silica gel, Clay, Activated charcoal.
Preparing the Formulation for Hard Gelatin Capsule
The active and inert ingredients should be thoroughly mixed to ensure a uniform mixing of the filling powder. It is advantageous when the densities and particle sizes of the drug and excipient are similar in order to achieve a uniform drug distribution.
Hard gelatin capsules are used to encapsulate approximately 65 mg to 1 g of powdered material, depending on the properties of the powdered material.
Manufacturing Process of Hard Gelatin Capsule Shells
Capsule shells are produced industrially by the mechanical dipping of pins or pegs of desired shape and diameter into a temperature controlled reservoir of melted gelatin mixture.
Steps involved in making empty gelatin capsules;
1. Dipping
2. Spinning or Rotation
3. Drying
4 Stripping
5 Trimming and Joining
6. Polishing
7. Printing
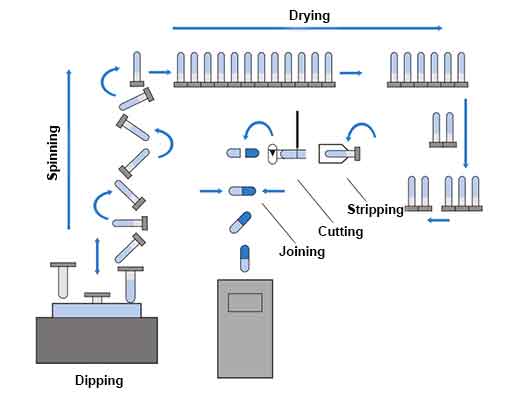
These process are described below;
1. Dipping
A pair of stainless steel pins is immersed in a dipping solution to form the cap and body at the same time. The dipping solution was maintained at a temperature of about 500 ° C. in a heated jacketed dipping pan.
2. Spinning or Rotation
The pins are rotated to evenly distribute the gelatin on the pins and to prevent the formation of a bead at the capsule ends.
3. Drying
Gelatin is dried with cool steam to form a hard crust. The pins move through a series of air drying kilns to remove water.
4. Stripping
A series of bronze jaws remove the capsule cap and body from the pins.

5. Trimming and Joining
The stripped cap and body parts are trimmed to the required length with a fixed knife. After being trimmed to the correct length, the cap and body part are combined and ejected from the machine. The finished capsules are pushed onto a conveyor belt and transferred to containers.
The quality of the capsules is controlled throughout the manufacturing process, including size, moisture content, wall thickness and color. Capsules are sorted and visually inspected in specially designed inspection stations.
6. Polishing
There are 3 types of polishing;
1. Pan Polishing: Acela-cota pan is used to remove dust & polish.
2. Cloth Dusting: Capsule are rubbed with cloth.
3. Brushing: Capsule are feed under soft rotating brush.
7. Printing
Perfect capsules are imprinted with the client logo.
Filling Of Hard Gelatin Capsules
There are three processes of filling of hard gelatin capsules. They are;
1. Punch Method or Manual Filling
2. Hand Operated methods
3. Automatic filling machine or Semi-Automatic Capsules Devices
1. Punch Method or Manual Filling
In this method precise number and size of capsule are selected which are to be filled. Then the capsules body and caps are separated. The powder to be encapsulated is taken and placed on a piece of clean paper or glass sheet or porcelain plate using spatula which is formed into a cake having a depth of approximately one-fourth to one-third the length of the capsule body.

Then empty capsule body is held between the thumb and forefinger and punched vertically into the powder cake repeatedly until filled.
2. Hand Operated methods
Hand operated and electrically operated machines are in practice for filling the capsules but for small and quick dispensing hand operated machines are quite economical.
Example of hand operated capsule filling machine is Feton Capsule Filling Machine.
A hand operated hard gelatin capsule filling machine consists of the following parts;
1. A bed with 200-300 holes.
2. A capsule loading tray
3. A powder tray
4. A pin plate having 200 or 300 pins corresponding to the number of holes in the bed and capsule loading tray.
5. A lever
6. A handle
7. A plate fitted with rubber top

All parts of the machine are made up of stainless steel. The machines are generally supplied with additional loading trays, beds, and pin plates with various diameters of holes so as to fill the desired size of the capsules.
These machine are very simple to operate, can be easily dismantled and reassembled.
Working Process of Hand Operated Machine
The empty capsules are filled into the loading tray which is then placed over the bed.
↓
By operating the handle, the bodies of the capsules are locked and caps separated in the loading tray itself which is then removed by operating the lever.
↓
The weighed amount of the drug to be filled in the capsules is placed in powder tray already kept in position over the bed.
↓
Spread the powder with the help of a powder spreader so as to fill the bodies of the capsules uniformly.
↓
Collect excess of the powder on the platform of the powder tray.
↓
Lower the pin plate and move it downward so as to press the powder in the bodies.
↓
Remove the powder tray and place the caps holding tray in position.
↓
Press the caps with the help of plate with rubber top and operate the lever to unlock the cap and body of the capsules.
↓
Remove the loading tray and collect the filled capsules in a tray.
With 200 hole machine about 5000 capsules can be filled per hour and with 300 hole machine 7500 capsules can be filled per hour.
3. Automatic filling machine or Semi-Automatic Capsules Devices
On large scale manufacturing various types of semiautomatic and automatic machines are used.
They operate on the same principle as manual filling, namely the caps are removed powder filled in the bodies, caps replaced and filled capsules are ejected out.

The process of working of automatic hard gelatin capsule filling machines are;
1. Rectification
Rectification means “the act of making straight or correct”. In this stage;
- The empty capsule shells are oriented in such a way so that they are all pointing in the same direction that is “body end downward”.
- The capsule pass one-at-a time, through a channel just wide enough to provide a frictional grip at the cap end.
- A special designed blade push against the capsule & causes it to rotate about its cap end as a fulcrum (as the fixed point on which the liver moves). After two pushes (one horizontally & one vertically downward) the capsule will always be aligned body end downward.
2. Separating the caps from empty capsules
3. Filling the bodies
4. Scraping the excess powder
5. Replacing the caps
6. Sealing the capsules
7. Cleaning the outside of the filled capsules
With automatic capsule filling machines powders or granulated products can be filled into hard gelatin capsules. With accessory equipment, pellets or tablets along with powders can also be filled into the capsules. Depending upon the make and model, operator and the type of the materials to be filled, a machine can fill 9000 to 150,000 capsules per hour. Various automatic machines which are available in market are;
1. Eli-lily and Co
2. Farmatic
3. Osaka
4. Hofliger and Karg
5. Zanasi Nigris
6. Parke-Davis
7. Macofar SAS
These machine differ in their design and output.
Sealing Of Hard Gelatin Capsules
In this process, the two capsule parts are sealed with a gelatin or polymer band at the joint of the cap and body.
A tamper-resistant seal on hard gelatin capsules was developed in which the contact areas of the cap and body are wetted with a mixture of water and ethanol to soften the gelatin and then thermally bonded at 40-45°C to form the seal.
2. Soft Gelatin Capsules
Soft gelatin capsules are one piece, hermetically sealed, soft gelatin shells containing liquids, suspensions, or semi-solids preparation.
Soft gelatin is mainly composed of gelatin, plasticizers, preservative, coloring and opacifying agents, flavoring agents and sugars.

Manufacturing Of Soft Gelatin Capsules
Soft Gelatin capsules are one piece, hermetically (completely airtight) sealed. The shape of soft gelatin capsule can be round, oval, oblong, or tube. Soft shell mainly composed of gelatin, plasticizers, preservative, coloring and opacifying agents, flavoring agents and sugars.
The ratio of dry plasticizer to dry gelatin determines the “hardness” of the shell and vary from 0.3-1.0 for very hard shell to 1.0-1.8 for very soft shell.
Up to 5% sugar may be included to give a “chewable” quality to the shell. The residual shell moisture content of finished capsules will be in the range of 6-10%. The pH of the liquid used can be range between 2.5 to 7.5.
Two types of vehicles used in soft gelatin capsules;
1) Water immiscible, volatile & nonvolatile liquids: Vegetable, aromatic oils & mineral oils, Aromatic & aliphatic hydrocarbons, Acetylated glycerin, esters, and organic acids.
2) Water miscible, nonvolatile liquids: Polyethylene glycols (PEG) with low molecular weight used more recently because of their ability to mix with water and accelerate dissolution of dissolved or suspended drugs, Nonionic surface active agents (Polysorbate 80) & Propylene glycol
Soft Capsules are manufactured by four methods. They are;
1. Plate Process
2. Rotary Die Process
3. Reciprocating Die
4. Accogel Machine
Among these process, Plate Process and Rotary Die Process are most common.
1. Plate Process
- In the plate process method, a heated sheet of plasticized gelatin is placed on a plate with several cavities or molds, and the sheet is drawn into these cavities by applying a vacuum.
- A measured quantity of liquid medicament is poured over it, then another sheet of gelatin is placed on it.
- Over this another plate of the mould is placed and the pressure is then applied to the combined plates.
- The capsules are then simultaneously formed, filled, sealed and cut into individual units.
This is the simplest and easiest technique among all the techniques of Soft gelatin encapsulation
2. Rotary Die Process
- In the rotary die process filled capsules are produced continuously and automatically.
- Two continuous gelatin sheets are supplied to the two die rolls of the machine which has a number of matching dies and rotate at the same speed and in the opposite direction.
- When the gelatin sheets come between the rollers, the material to be filled is injected through a measuring device.
- The pressure created by the material forces the gelatin sheet to enter in the cavities of the die rolls to form two halves of the capsule and fill them.
- The heat and pressure generated by the die rolls seals and cuts out the capsules.
- The finished capsules are then passed through a series of naphtha baths to remove lubricants and then dried.
- These rotary die machines can produce 25,000 to 30,000 capsules per hour.

Drying, Cleaning, Polishing and Storage of Soft Gelatin Capsules
After the soft gels are formed, they contain around 20% of water, which must be dried to maintain its integrity.
On a small scale, capsules cleaned individually by rubbing them with a clean gauze or cloth.
On a large scale, capsule-filling machines are affixed with a cleaning vacuum that removes any extraneous material from the capsules as they exit the equipment.
Cleaning and polishing of the soft gelatin capsule is done to remove any mineral oil or glycerin which may adhere on the outer skin of the capsules.
Soft gelatin capsule product should be stored in the temperature around 20-24⁰C.
Differences between Hard Gelatin Capsules and Soft Gelatin Capsules
| Hard Gelatin Capsules | Soft Gelatin Capsules |
| Two pieces (large body & short cap) | One piece & hermetically sealed |
| Cylindrical in shape | Available in round , oval & tube like shapes |
| Powder drug or pallets coated with drug are encapsulated. | Liquid & semi liquid fill & unstable substances are encapsulated |
| Gelatin in hard form is used | Molten gelatin are used |
| Capsules sealed after filled to ensure that medicaments not come out of capsule due to rough handling | Filling & sealing of soft gelatin capsules are done in a combined operation on machine |
| Have 8 different type of sizes | No specific size |
Packaging and Storage of Capsules
Capsules should be packed in well closed glass or plastic containers and stored in a cool place. These types of containers have advantage over cardboard boxes that they are more convenient to handle and transport and protect the capsules from moisture and dust. To prevent the capsules from rattling a tuft of cotton is placed over and under the capsules in the vials. In vials containing very hygroscopic capsules a packet containing desiccant like silica gel or anhydrous calcium chloride may be placed to prevent the absorption of excessive moisture by the capsules. Now a days capsules are strip packaged which provide sanitary handling of medicines, ease in counting and identification. Soft gelatin capsule product should be stored in the temperature around 20-24⁰C.
Standardization of Capsules
Whether capsules are produced on a small or large scale all of them are required to pass not only the disintegration test, weight variation test and percentage of medicament test but a visual inspection must be made as they roll off the capsule machine onto a conveyor belt regarding uniformity in shape, size, colour and filling. As the capsules move in front of the inspectors the visibly defective or suspected of being less than perfect are picked out. If the number of defective capsules is large it may be due to some fault in the capsule filling machine which must be corrected. The hard and soft gelatin capsules should be subjected following tests for their standardization:
1. Shape and size.
2. Colour.
3. Thickness of capsule shell.
4. Leaking test for semi-solid and liquid ingredients from soft capsules.
5. Disintegration test.
6. Weight variation test.
7. Percentage of medicament test.
In official books the following quality control tests are recommended for capsules:
(a) Disintegration Test
For performing disintegration test on capsules the tablet disintegration test apparatus is used but the guiding disc may not be used except that the capsules float on top of the water.
One capsule is placed in each tube which are then suspended in the beakers to move up and down for 30 minutes.
The capsules pass the test if no residue of drug or other than fragments of shell remains on No.10 mesh screen of the tubes.
(b) Dissolution Test
Place 1000 ml of water free from dissolved air having temperature of 36.5ºC to 37.5ºC. After that place specified number of capsules in each basket. Start motor and adjust speed 100 rpm.
Withdraw the required volume of solution after 45 minutes. Then Filter and weigh the amount of active ingredients by the method specified.
Repeat the test for four times. The test is said to pass if the amount of active ingredient is not less than 70% of the stated amount
(c) Weight Variation Test
20 capsules are taken at random and weighed. Their average weight is calculated. Then each capsule is weighed individually and their weight noted.
The capsules pass the test if the weight of individual capsule falls within 90-110% of the average weight.
If this requirement is not met, then the weight of the contents for each individual capsule is determined and compared with the average weight of contents.
The contents from the shells can be removed just by emptying or with the help of small brush.
From soft gelatin capsules the contents are removed by squeezing the shells which has been carefully cut.
The remainder contents are removed by washing with a suitable solvent.
After drying the shells, they are weighed and the content weights of the individual capsules are calculated. The requirements are met if;
(a) Not more than 2 of the differences are greater than 10% of the average net content and
(b) In no case the difference is greater than 25%.
(d) Contents Uniformity Test
This test is applicable to all capsules which are meant for oral administration. For this test a sample of the contents is assayed as described in individual monographs and the values calculated which must comply with the prescribed standards.
In this test randomly 30 tablets are selected and 10 of these assayed individually, if 9 out of 10 within the requirement of ± 15% (85%-115%) limits and the tenth tablet is not outside ± 25% (75%-125%) so the batch should be accepted.
If 2 capsules out of 10 fall outside the range ± 15%, then take 20 capsules and assay them individually and take the average of 30 capsules. The batch should be accepted, if 27 capsules in the ± 15% range and only 3 capsules in the ± 25% range.
